EXOMIND | CASE STUDY
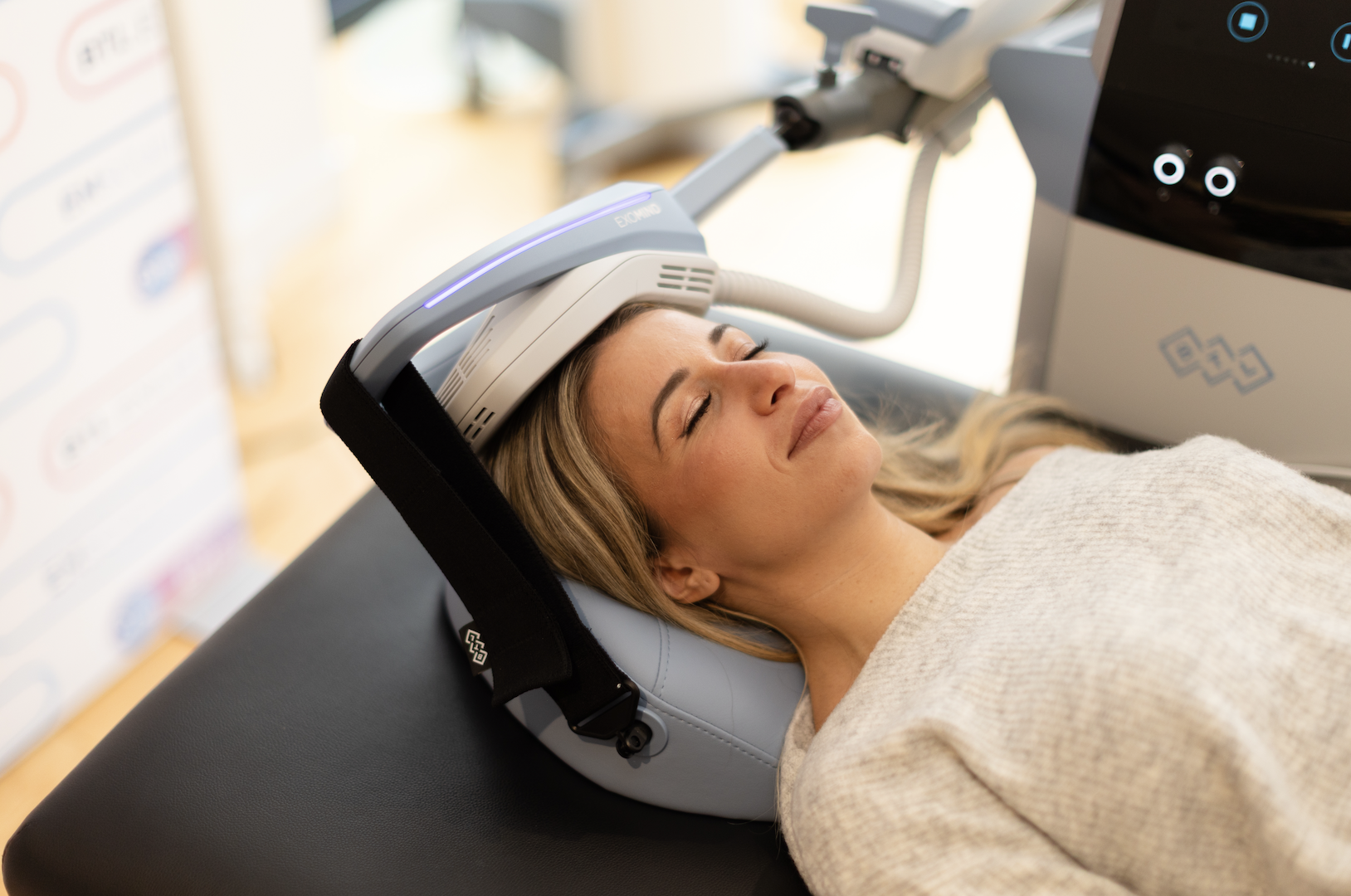
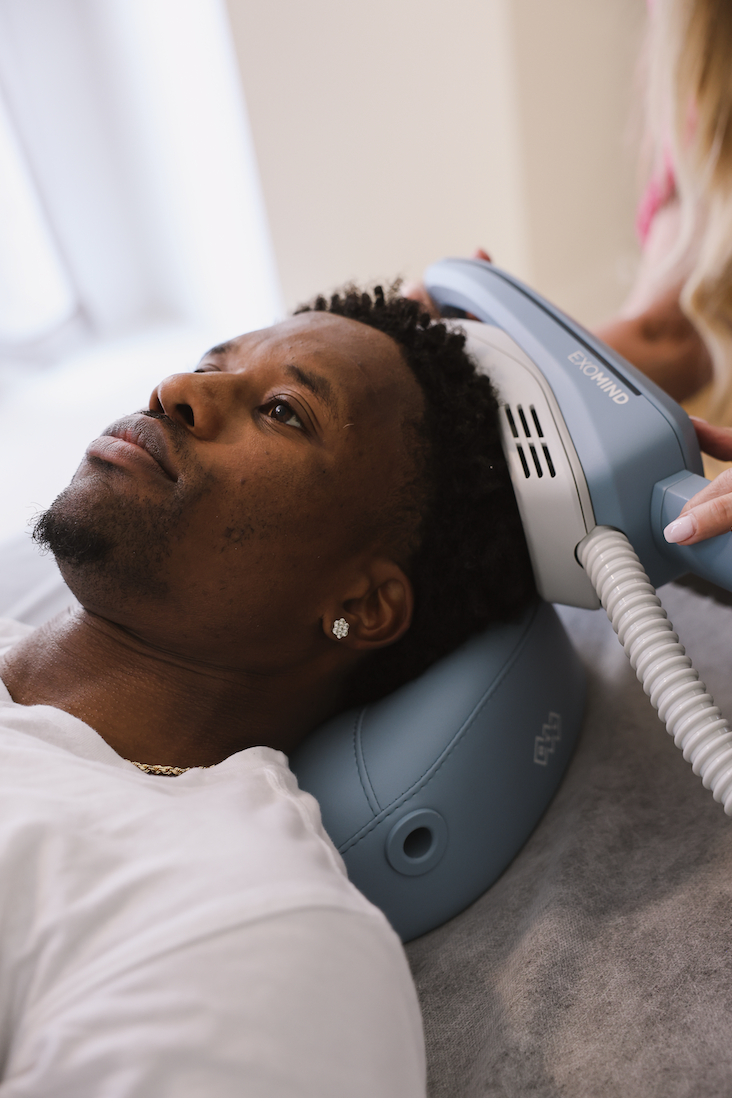
In early 2025, ssp launched BTL’s FDA-cleared Exomind, the next-generation transcranial magnetic stimulation (TMS) treatment that helps patients enhance their neuroplasticity, strengthening the brain’s ability to cope, adapt and stay resilient.
Spearheading the emerging demand for “neuro” treatments and growing focus on mental wellness, we positioned Exomind as the next-generation treatment to help patients manage stress, depression and anxiety, treat perimenopausal brain fog, and reduce cortisol-driven skin conditions like acne and eczema by translating complicated scientific and medical data into accessible, relatable storytelling and positioning our suite of early adapter MDs as thought leaders in the media.
In less than six months, we generated extensive worldwide buzz, demand and awareness via a multitude of high-profile press coverage in outlets including Vogue, Elle, Time, Air Mail and The LA Times, and successfully identified and treated key influencers, KOLs and celebrities struggling with relatable conditions. These results empowered BTL’s global sales teams to reach high-profile providers and open hundreds of new accounts, and drove patients to ask for Exomind by name during wellness appointments with their providers.
To date, Exomind sold more units in one quarter than its closest competitor sold in all of 2025.
“Working with ssp has been a step-change. We saw more meaningful visibility in a single month than we saw in the entire previous year with another firm.”
~ John Ferris, VP Marketing, BTL
< CLIENTS